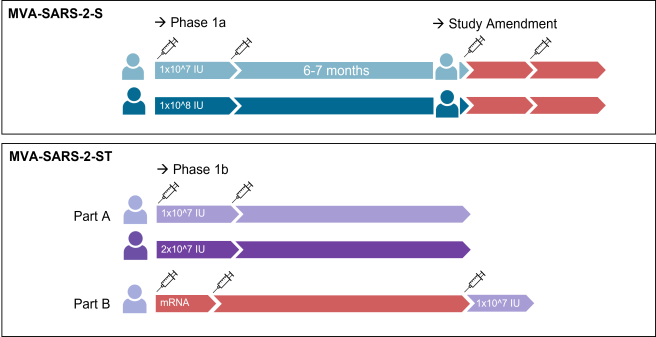
Vaccine Trial: MVA-SARS-2-S
An open, single-center Phase I trial to assess the safety, tolerability and immunogenicity of two ascending doses of the candidate vaccine MVA-SARS-2-S and heterologous booster vaccinations with a licensed vaccine against COVID-19.
Clinical Trial
database
End of September 2020, the University Medical Centre Hamburg-Eppendorf (UKE) received approval from the Paul-Ehrlich-Institut, Federal Institute for Vaccines and Biomedical Drugs, and the Ethics Commission of the Hamburg Medical Association to start clinical trials. On October 9, the first subject was injected with the vaccine MVA-SARS-2-S against COVID-19. The results were available in January 2021: The vaccine proved to be safe, but the effect fell short of expectations.
DZIF